|
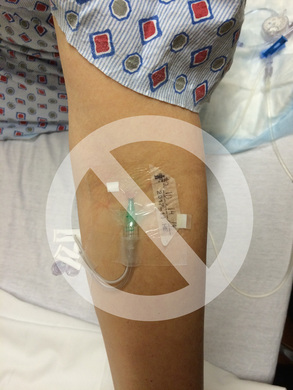
For many years, we have overlooked the infection risk associated with peripheral IV (PIV) catheters while focusing exclusively on infection risk associated with central vascular access devices. Our attention must include both! While there is no doubt that published rates of central line associated bloodstream infection (CLABSI) were far too high, we must take a closer look at the rates of BSI that are associated with PIV catheters as well. Unfortunately, there is very little prospective data on actual PIV-BSI rates. There are 2 unique studies on Staphylococcus aureus BSI that used retrospective data to analyze the problem. These studies reported similar processes and outcomes. Both studies used medical records to identify patients with S. aureus BSI and correlated blood culture results with clinical data. Trinh, et.al, reported 24 cases of S. aureus BSI related to PIV catheters. They reported these patients to be more likely to have the PIV inserted in the emergency department or outside the hospital and to have the PIV placed in the antecubital fossa. Of all S. aureus BSI, 12% were caused by PIV catheters. Using this data and data on all US hospital adult discharges, these researchers estimated that as many as 10,000 PIV-BSIs from S. aureus alone happen in US hospitals. Austin, et.al, reported 34 of 445 (7.6%) of all S. aureus BSI were related to thrombophlebitis at a PIV catheter site. The length of the bacteremic period was longer in patients with PIV-BSI, primarily caused by methicillin-resistant S. aureus BSI. Most cases were caused by PIV catheters inserted in the proximal forearm and antecubital fossa. Also, most cases were due to thrombophlebitis at old PIV sites. No guidelines recommend frequent monitoring of previous PIV catheter sites for developing or worsening signs and symptoms of thrombophlebitis, however this study indicates the necessity for monitoring sites after PIV removal. The bottom line is the need for careful aseptic technique with each PIV catheter insertion. Don’t use veins of the antecubital fossa. One or two swipes on the skin with an alcohol pad is not sufficient. Touching the prepped site with gloved fingers can only be done when wearing sterile gloves. All methods of infection prevention must be used. Hospitals are no longer reimbursed for treating infections associated with any type of vascular access device, including PIV-BSI. Strict attention to PIV catheter insertion and maintenance is just as important as the attention we now give to central VADs. Short Peripheral Intravenous Catheters and Infections Trinh, T. T., Chan, P. A., Edwards, O., Hollenbeck, B., Huang, B., Burdick, N., . . . Mermel, L. A. (2011). Peripheral Venous Catheter-Related Staphylococcus aureus Bacteremia. Infection control and hospital epidemiology: the official journal of the Society of Hospital Epidemiologists of America, 32(6), 579. Austin, E. D., Sullivan, S. B., Whittier, S., Lowy, F. D., & Uhlemann, A.-C. (2016). Peripheral intravenous catheter placement is an underrecognized source of Staphylococcus aureus bloodstream infection. Paper presented at the Open forum infectious diseases. Online Course Updated - Peripheral IV Therapy in Adults One of our most popular courses has been updated with content that incorporates the 2016 Infusion Therapy Standards of Practice. Start out on the right track with this basic course. If you are new to IV therapy, this course will provide the needed information about all steps in process of patient assessment, planning what is needed, performing the procedures and assessing the outcomes. Click here to learn more! |
Author: Lynn HadawayLynn Hadaway is an international thought leader in infusion therapy and vascular access, having been in this practice for more than 40 years. Her experience comes from hospital-based infusion teams, device manufacturers, and continuing education services. Her journal and textbook publications are extensive. She also maintains board certification in infusion nursing (CRNI) and nursing professional development (RN-BC). Categories
Archives
May 2019
|
Copyright © 2020 Lynn Hadaway Associates, Inc. | P.O. Box 10, Milner, GA 30257 | 770-358-7861


 RSS Feed
RSS Feed