|
Yes, the parameters for pH have been removed from the INS Standards of Practice. Evidence shows that this is not the critical factor in reducing intraluminal vein damage that we originally thought it to be. But I have encountered lots of confusion on this issue since the Standards were released.
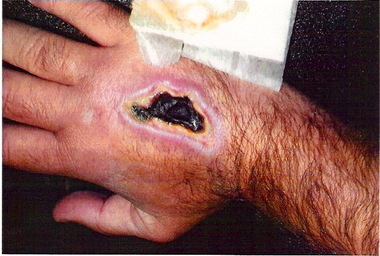
There seems to be a prevailing idea that pH of the solution is the only factor that causes a medication to be a vesicant but this is not a correct idea. In our online course, Infiltration and Extravasation from Vascular Access Devices, 5 causes of tissue damage are discussed including osmotic pressure, non-physiologic pH, ischemia, mechanical compression, and cellular toxicity. Additionally, an I.V. medication can cause vein irritation (phlebitis) and subcutaneous tissue damage due to the excipients in the medication. Excipients are the “inactive ingredients” added to medications, primarily for the sake of drug stability. Examples include various types of alcohol, preservatives, and other agents. Here is the full ingredient list for promethazine: “Each mL contains promethazine HCl, either 25 mg or 50 mg, edetate disodium 0.1 mg, calcium chloride 0.04 mg, sodium metabisulfite 0.25 mg and phenol 5 mg in Water for injection. pH 4.0 to 5.5; buffered with acetic acid-sodium acetate.” The full ingredient list for phenytoin sodium injection (Dilantin) is: “phenytoin sodium in a vehicle containing 40% propylene glycol and 10% alcohol in water for injection, adjusted to pH 12 with sodium hydroxide.” Vancomycin may contain hydrochloric acid and/or sodium hydroxide for pH adjustment. Nafcillin has dextrose, sodium citrate hydrous, hydrochloric acid or sodium hydroxide added for a final pH between 6.5 to 8. Although the pH of nafcillin is as close as any drug to physiologic pH, the drug literature also states: “Pain, swelling, inflammation, phlebitis, thrombophlebitis, and occasional skin sloughing at the injection site have occurred with intravenous administration of nafcillin. (See Dosage and Administration.) Severe tissue necrosis with sloughing secondary to subcutaneous extravasation of nafcillin has been reported.” The list of non-antineoplastic medications that are vesicants is extensive and also included in our online course. “Vesicant” is defined as a drug causing blisters and other tissue injury that may be severe and can lead to tissue necrosis. There is nothing in this definition about the actual reason for any drug being a vesicant. For this reason, anyone involved with administering I.V. medications must know the full nature of these medications, and the risks involved. Infiltration and extravasation are one of the most common causes of infusion-related lawsuits. Learn more by taking our online course. Click here for more information.
Judy Comstock
3/24/2016 11:14:32 pm
Wondering if you ever present on this topic for Ava Network meetings?
Lynn Hadaway
3/30/2016 05:19:43 am
Hi Judy, yes I do present a variety of topics at many chapter meetings for any professional organization. This can be done in person or by use of a videoconference. In person is a higher fee plus travel expenses. The videoconference presentation is much less expensive. Please contact me by email at lynn@hadawayassociates.com or by phone at 770-358-7861 to discuss your needs. Comments are closed.
|
Author: Lynn HadawayLynn Hadaway is an international thought leader in infusion therapy and vascular access, having been in this practice for more than 40 years. Her experience comes from hospital-based infusion teams, device manufacturers, and continuing education services. Her journal and textbook publications are extensive. She also maintains board certification in infusion nursing (CRNI) and nursing professional development (RN-BC). Categories
Archives
May 2019
|
Copyright © 2020 Lynn Hadaway Associates, Inc. | P.O. Box 10, Milner, GA 30257 | 770-358-7861


 RSS Feed
RSS Feed