|
Yes, the parameters for pH have been removed from the INS Standards of Practice. Evidence shows that this is not the critical factor in reducing intraluminal vein damage that we originally thought it to be. But I have encountered lots of confusion on this issue since the Standards were released.
There seems to be a prevailing idea that pH of the solution is the only factor that causes a medication to be a vesicant but this is not a correct idea. In our online course, Infiltration and Extravasation from Vascular Access Devices, 5 causes of tissue damage are discussed including osmotic pressure, non-physiologic pH, ischemia, mechanical compression, and cellular toxicity. Additionally, an I.V. medication can cause vein irritation (phlebitis) and subcutaneous tissue damage due to the excipients in the medication. Excipients are the “inactive ingredients” added to medications, primarily for the sake of drug stability. Examples include various types of alcohol, preservatives, and other agents. Here is the full ingredient list for promethazine: “Each mL contains promethazine HCl, either 25 mg or 50 mg, edetate disodium 0.1 mg, calcium chloride 0.04 mg, sodium metabisulfite 0.25 mg and phenol 5 mg in Water for injection. pH 4.0 to 5.5; buffered with acetic acid-sodium acetate.” The full ingredient list for phenytoin sodium injection (Dilantin) is: “phenytoin sodium in a vehicle containing 40% propylene glycol and 10% alcohol in water for injection, adjusted to pH 12 with sodium hydroxide.” Vancomycin may contain hydrochloric acid and/or sodium hydroxide for pH adjustment. Nafcillin has dextrose, sodium citrate hydrous, hydrochloric acid or sodium hydroxide added for a final pH between 6.5 to 8. Although the pH of nafcillin is as close as any drug to physiologic pH, the drug literature also states: “Pain, swelling, inflammation, phlebitis, thrombophlebitis, and occasional skin sloughing at the injection site have occurred with intravenous administration of nafcillin. (See Dosage and Administration.) Severe tissue necrosis with sloughing secondary to subcutaneous extravasation of nafcillin has been reported.” The list of non-antineoplastic medications that are vesicants is extensive and also included in our online course. “Vesicant” is defined as a drug causing blisters and other tissue injury that may be severe and can lead to tissue necrosis. There is nothing in this definition about the actual reason for any drug being a vesicant. For this reason, anyone involved with administering I.V. medications must know the full nature of these medications, and the risks involved. Infiltration and extravasation are one of the most common causes of infusion-related lawsuits. Learn more by taking our online course. Click here for more information.  The typical procedure for thrombolysis in central vascular access devices calls for the use of tPA (Alteplase) 2 mg in 2 mL. But what about the implanted ports that hold more volume than 2 mL? The internal volume of the catheter plus the port body plus the access needle could easily be more than 2 mL. Large lumen catheters and high profile port bodies could easily equal 3 mL or more. Before using any medication for catheter clearance, ensure you have ruled out other causes of the occlusion. VADs without a blood return should not be used, but make sure your technique is not the problem. Use a slow, gentle technique to aspirate. Use a smaller diameter syringe (e.g. 5 mL or 3 mL) as these cause LESS pressure on aspiration. Look at the history of the problem to determine if this is truly a thrombotic occlusion or if it could be caused by contact between incompatible medications producing a precipitate or a total nutrient admixture that causes a lipid buildup on the intraluminal catheter walls. Mechanical problems such as pinch-off syndrome should also be considered for subclavian insertion sites. Also check for external problems such as closed clamps or kinked tubing. The internal volume of the entire catheter system can be difficult to determine. The design of the port body and lumen size of the catheter varies. The manufacturer provides the internal volume or priming volume of each brand of implanted port. However, the catheter is cut to a patient-specific length, which decreases the original internal volume. Several interventions to manage this problem should be considered. Obtaining the internal lumen volume could be done by simple aspiration. First flush the catheter with normal saline. Attach an empty syringe, and slowly aspirate until the blood has reached the connection of the syringe to the needleless connector on the port access extension set. Note the space in the syringe as this should approximate the catheter’s internal volume. But remember, this has to be done and documented before an occlusion occurs. This volume would determine the amount of tPA required to fill the entire length of the system. The other option is to instill the tPA dose of 2 mg in 2 mL, and then follow it be a small amount of normal saline such as 0.5 to 1 mL to move the tPA to the distal end of the catheter and into direct contact with the location of the thrombus. Dilution of the tPA to less than 2 mg in 2 mL by adding more volume is not recommended as this may not be as effective. I would recommend reading the 2016 Infusion Therapy Standard of Practice #48 Central Vascular Access Device Occlusion for more details. This question is not addressed because there are no studies reporting these variations, but there can be concern among nurses doing this procedure for implanted ports. There are currently several midline catheters available with a labeled indication for power injection of contrast agents. While the catheter are build to withstand the pressure that may be created with power injection, we need to ask another question – Is it safe to use a midline tip location for power injection of contrast agents? The simple answer is we don’t know yet.
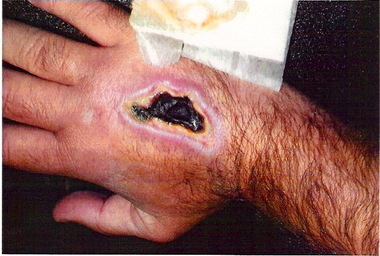
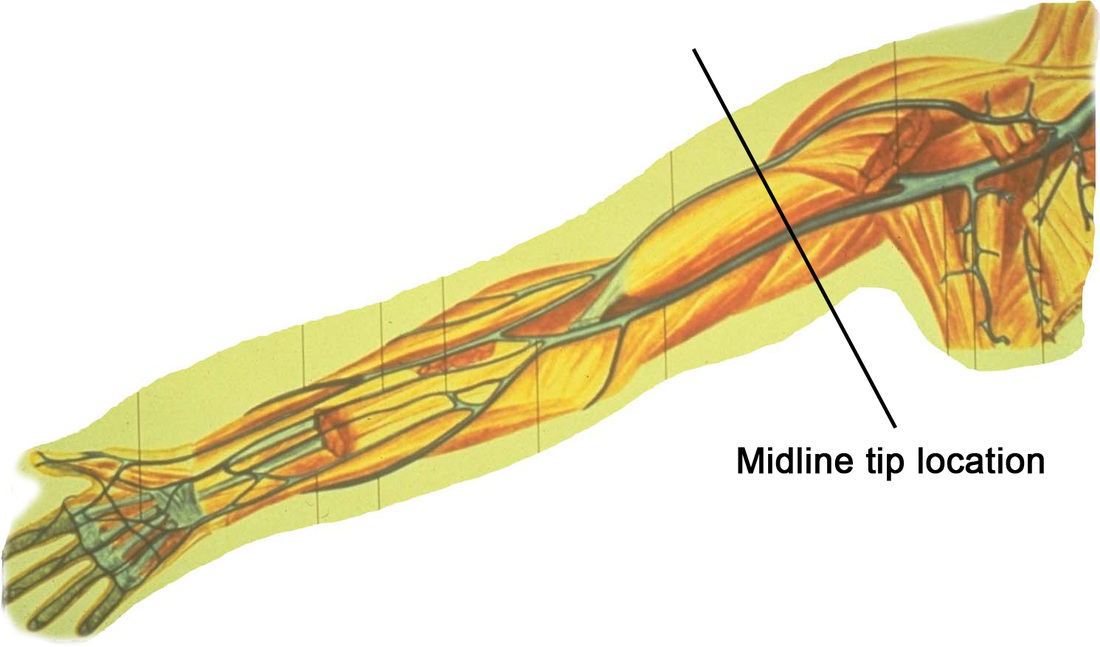
The concern is the injection of a hypertonic contrast agent considered to be vesicants into the deep upper arm location of a midline catheter. Deep veins could mean that fluid in the tissue may go undetected and potentially damage large arteries and nerves in the area. Many clinicians report that a midline catheter will not typically produce a blood return, thus eliminating one component of a complete clinical assessment of catheter patency. One the positive side, evidence is showing that longer catheters are needed when ultrasound is used to access deep vessels. Higher rates of infiltration/extravasation are reported when deep veins are used, even with 1.8 and 2 inch long catheters. This may indicate tht a 3 inch long catheter could be safer than a 2 inch catheter in the veins of the upper extremity. At the present time, there is a distinct need for more research on midline catheters and the midline tip location. I am aware of 2 simple descriptive clinical studies on the newer midline catheters labeled for power injection. These studies included a brief statement about small numbers of patients having the midline catheter used for contrast injection. These studies did not include details of the types of contrast agents used, the rates of injection, or the dwell time of the midline catheter when the contrast was injected. Extravasation with contrast agents happens, although it is reported to have very low rates usually less than 1%. In my experience as an expert witness, I have seen about 15 cases of contrast extravasation however all have involved short peripheral catheters inserted in the hand, wrist or antecubital fossa. No cases have involved a midline catheter or midline tip location. The bottom line is we need more data for these critical clinical decisions.  I think that the language we use is important for adequate communication. If we are not using the same words to indicate certain actions, how can we understand each other? Or adequately communicate good practices to our patients? The 2016 Infusion Therapy Standards of Practice contains an expanded Glossary so we can enhance our communication with each other. One area where there seems to much confusion is how we use the words “flushing” and “locking” for all VADs. The INS glossary defines
Flushing is done most commonly with normal saline, although a few drugs may be incompatible with saline. In that case, 5% dextrose in water is used to flush the drug from the lumen but saline should be used to flush out the dextrose as this will provide nutrients for growing biofilm. Solutions for locking a VAD are expanding. Evidence now shows that either normal saline or heparin lock solution produces similar outcomes for locking a central VAD. One solution is not better than the other. Additionally, new antimicrobial lock solutions such as ethanol, citrate, and numerous antibiotic solutions are proving to prevent and treat catheter related bloodstream infection. All studies refer to these as “locking solutions”. Different purposes, a variety of solutions, different volumes, and locking periods makes this an expanding and complex topic. So flushing and locking are not interchangeable terms. Using the correct word will enhance interprofessional communication.  The 2016 Infusion Therapy Standards of Practice have already generated questions about one hot issue – should short peripheral catheters be routinely aspirated for a blood return to assess their patency? For the first time, this document includes a definition of “blood return” in the glossary, stating this is a component of VAD patency assessment and that the blood should be the color and consistency of whole blood. Blood return from peripheral catheters is included in standards on documentation, flushing and locking, VAD assessment, VAD removal, infiltration and extravasation, and antineoplastic agents. Many old nursing references state that reliance on blood return is not adequate to determine if the catheter is infiltrating fluid. The nurse can not rely exclusively on the presence of absence of a blood return before using the peripheral catheter. There could easily be a second puncture in the posterior vein wall and still get a blood return OR there could be a small catheter occluded by aspiration technique. For these reasons, the assessment must also include observing the appearance of the site, including comparison to the opposite extremity; palpating for change in temperature or induration; and patient complaints about the site. Other reasons for a lack of blood return could be damage to the endothelial vein lining producing edema or small thrombi that occlude the back flow of blood. Fluid may not be escaping from the vein – yet. But this damage can lead to infiltration or extravasation and continued use will only exacerbate the current complication. Complications are seen more frequently when venous sites in the hand, wrist, and antecubital fossa are used. The 2016 Standards now call for obtaining a blood sample from a peripheral catheter during the dwell time instead of during the insertion procedure. Studies are showing that this is a successful practice. If the peripheral catheter will yield a blood sample, it can easily produce a blood return for patency assessment. As I mentioned, technique can be the cause of failure to obtain a blood return. First and foremost, use a slow and gentle technique to aspirate from the catheter. Pulling hard and fast on the syringe plunger can pull the vein wall over the catheter lumen. Next, change to a smaller syringe as aspiration with a smaller syringe produces less pressure and better results. That is opposite from what happens with injection where a larger syringe is recommended. Finally, put a tourniquet on the arm above the catheter tip to see if there is a blood return. If all of these techniques fail to produce a blood return, do NOT use the catheter. Everyone can think of many examples of complications associated with vascular access devices and infusion therapy. Textbooks and all types of journal publications provide much information about the signs and symptoms, prevention, and management of these events - always with the goal of prevention.
What about those adverse events that are not found in the published literature? Do they actually happen? If a study or case report about a specific complication can not be found in the published literature should we dismiss the idea that it could happen? A primary example is venous air embolism (VAE) following the removal of a PICC. The catheter is slowly withdrawn from the vein and pressure is applied to stop any bleeding. Do we need to use the same precautions to prevent VAE with a PICC removal as we use with a subclavian or jugular insertion site? After searching extensively through the published literature in the English language, there are no publications that have documented VAE with PICC removal. There are a few reports of VAE with PICCs occurring due to attaching an unprimed administration set but none occurring during PICC removal. Based on anatomy and physiology, we know that a skin-to-vein tract can develop especially with lengthy catheter dwell times. Also, a fibrin sheath frequently covers the entire length of catheter and could remain intact inside the vein when the catheter is removed. The 2016 Infusion Therapy Standards of Practice includes this information and advises to use methods to prevent VAE for all central vascular access devices (CVAD). In my opinion, we must use our knowledge of vascular anatomy and the physiology of blood flow, merged with our critical thinking skills based on patient assessment. Armed with this information, our professional responsibility is to apply the safest methods possible to prevent such events. Additionally, if an unreported event occurs in your facility, please share your experience by publishing your case report. No publications on a complication, yet anatomical and physiological evidence supports the possibility indicates the need to apply the usual precautions in this example. What is your opinion? Many evidence-based documents are now available to guide your practice decisions. The 2016 Infusion Therapy Standards of Practice are now available from INS. ISMP issued a new guidelines document on I.V. Push Medications in 2015. APIC recently issued a new CLABSI Implementation Guide and the SHEA Compendium on CLABSI was updated in 2014. Add documents from ONS, ASPEN, AVA, and many others. Do you have questions about the content of a specific document? Concern about conflicting information between these documents? Issues with implementing these documents into your clinical practice? We can help! Join our monthly videoconference to participate in discussions to get your questions answered. This will not be a formal presentation followed by a short question and answer session. We want to meet your needs in a different way. Choose a date from the following list and register. Each session is planned for Thursday. Send your questions via email to Lynn Hadaway at lynn@hadawayassociates.com, preferably by Monday before the session. Lynn will organize the questions and facilitate the discussion. At the scheduled date and time, follow the directions for connecting your computer and join this discussion. It is that simple! Lynn is an excellent resource to facilitate this discussion. She has been an author of the INS standards for the 2006, 2011, and 2016 edition. She was a part of the consensus group that produced the ISMP paper and worked on the APIC and SHEA documents. Do not miss this opportunity to get your questions answered. Regular price - $195 - unlimited number of participants in one location with one Internet connection 20% discount in honor of I.V. Nurses Day - January 25 thru 31- $175 Sign up now! Paresthesia is defined as “a sensation of tingling, tickling, pricking, or burning on the skin with no apparent physical cause.” This word is found in the literature on nerve injury.
The 2016 Infusion Therapy Standards of Practice, due to be available by the end of January includes a new standard on Nerve Injury. This new standard uses “paresthesia” to describe this type of pain that can occur during venipuncture or associated with infiltration/extravasation. During catheter insertion, complaints of electrical shock-like pain going in either direction on the arm, tingling, feelings of “pins and needles” or numbness is an indication that a nerve has been damaged by the needle. Nerves and veins are located very close together, making it nearly impossible to choose a venipuncture site without some risk of nerve injury. The new standard will contain a list of nerves and anatomical locations where this problem is most frequently reported. This event must be managed by carefully removing the needle and finding another location for the venipuncture. The chance of permanent nerve damage and subsequent complex regional pain syndrome (CRPS) increases when the inserter ignores these patient complaints and continues with the procedure in the same location. Pay careful attention to the type of pain complaints during all venipuncture procedures and never assume that this is the typical discomfort felt with most venipunctures. Paresthesia may also occur when excessive fluid is accumulating in the tissue. Nearby nerves are compressed causing these sensations. The fluid could be infusing I.V. fluid, blood from hematoma formation, or edema from inflammatory processes such as thrombophlebitis. Again, this is a reason for immediate catheter removal. Failure to remove the catheter quickly also increases the risk of permanent nerve damage and CRPS. The bottom line is paying careful attention to the patient’s complaints, asking questions about the type of pain, and taking quick action to reduce the risk of nerve damage. Read this new INS standard when it is available to learn more details. Additionally, investigate our comprehensive online education course on Infiltration and Extravasation. Details are located here. Today we are announcing dates and times for 2 new videoconferences: Antimicrobial Locking Solutions for Central Vascular Access Devices (CVAD) New evidence strongly supports the use of antimicrobial locking solutions to reduce unacceptably high rates of central line associated bloodstream infection (CLABSI). The 2016 Infusion Therapy Standards of Practice will address this new evidence with an expanded standard on flushing and locking. There is still a search for the most effective CVAD locking solution as this good news about CLABSI reduction comes with some obstacles. Before making clinical practice decisions, we invite you to participate in this discussion of the various locking solutions used, along with the good and not-so-good aspects of their use. Click here for scheduled dates, times, and registration information. Infection Prevention in Infusion Therapy: The Unanswered Questions Work on the 2016 Infusion Therapy Standards of Practice and other recent clinical guidelines revealed the absence of evidence for many critical questions about effective methods of infection prevention in infusion therapy. Recommendations for practice are supported by well-designed research, but many practices do not have supporting evidence. The problem could be very old studies that no longer reflect current practice, the total lack of evidence on specific questions, or be limited to in-vitro studies that are not easily applied to clinical practice. In the absence of evidence, how should you make decisions about these important aspects of clinical practice? This presentation was originally given at the 2015 annual conference of the Association for Practitioners in Infection Control ad Epidemiology (APIC). It was followed by a lively discussion then and in each videoconference. Click here for scheduled dates, times, and registration information. All videoconferences are scheduled for 90 minutes, which allows time for questions, answers, and discussion after the presentation. Gather all interested colleagues and join us for these presentations. We are limited in the number of computers that can sign on, so register early to reserve your spot. There is no limit on the number of people at your location.
 A few days ago, I received a question about confirming tip location of a newly inserted CVAD when the patient is located in the OR for insertion. As stated in many standards and guidelines documents, some method to confirm correct tip location is necessary before infusion begins through any CVAD. Historically, the most common method for this tip confirmation is by obtaining a chest xray. From the question I received, it would appear that obtaining a chest xray in the OR after a CVAD insertion is not common practice by surgeons and anesthesiologist. I can understand that adherence to the OR schedule is a significant issue and that time to obtain and interpret a chest xray may not be factored into that schedule. I have also heard discussions of insertion sites and malpositioned CVADs with some thinking that the internal jugular site has virtually no chance for malposition on insertion while others insertion sites have a greater risk. Many report that a chest xray is performed after the patient has been transferred to PACU or ICU, however this is after the catheter has been used for fluid and medication infusion during the surgical procedure. In my opinion, this sounds like a double standard – one for the OR and a different one for other patient care areas. While I cannot quote any data on what is the most common practice regarding this issue, I can point to the most recent set of guidelines from the American Society of Anesthesiologists. Click the title to gain free access to download Practice Guidelines for Central Venous Access: A Report by the American Society of Anesthesiologists Task Force on Central Venous Access was published in 2012 and provides valuable information on this issue. The document contains many valuable statements about preventing infection and other mechanical complications. The most important statement for the question at hand is: “The consultants and ASA members agree that a chest radiograph should be performed to confirm the location of the catheter tip as soon after catheterization as clinically appropriate. They also agree that, for central venous catheters placed in the operating room, a confirmatory chest radiograph may be performed in the early postoperative period. The ASA members agree and the consultants strongly agree that, if a chest radiograph is deferred to the postoperative period, pressure waveform analysis, blood gas analysis, ultrasound, or fluoroscopy should be used to confirm venous positioning of the catheter before use.” In the ASA statement above, pressure waveform and blood gas analysis will rule out arterial placement but it will not confirm where the tip is located. Numerous veins are identified as sites for malpositioned, suboptimal, or aberrant locations for a CVAD tip and these locations are known for increasing the risk of complications. One other issue with the ASA guideline is the allowance of tip locations in veins distal to the vena cava or cavoatrial junction. This position is in opposition to other guidelines stating the tip location should be located in the area extending from the distal superior vena cava to the upper right atrium with the most preferred site at the cavoatrial junction. For femoral insertion sites, the tip location should be above the diaphragm in the inferior vena cava at or near the cavoatrial junction. New technologies such as ECG and ultrasound are growing in application to confirm tip location with less reliance on a chest xray. ECG to confirm tip location, a practice this is prevalent in Europe, uses the stylet wire inside the catheter as an internal lead. When the wire reaches the SA node, the P wave is at is tallest point. This method works on most patients except those with cardiac anomalies that prevent a visible P wave from being detected on the ECG tracing. Ultrasound for tip location requires a special probe, the ability to locate the proper “acoustic window” for each patient, and the ability to interpret the findings. As we move toward implementation of these technologies, our goal should be to standardize practice across all locations. What is correct tip location and its confirmation should be the same for all patients in all healthcare facilities and departments. |
Author: Lynn HadawayLynn Hadaway is an international thought leader in infusion therapy and vascular access, having been in this practice for more than 40 years. Her experience comes from hospital-based infusion teams, device manufacturers, and continuing education services. Her journal and textbook publications are extensive. She also maintains board certification in infusion nursing (CRNI) and nursing professional development (RN-BC). Categories
Archives
May 2019
|
Copyright © 2020 Lynn Hadaway Associates, Inc. | P.O. Box 10, Milner, GA 30257 | 770-358-7861



 RSS Feed
RSS Feed